Hospital clinical laboratories form the foundation for accurate diagnosis, efficient workflows, and personnel safety in modern healthcare. This guide covers the core planning and design principles, international standards, floor plans, ventilation, electrical systems, plumbing, finishes, and key requirements for hospital clinical laboratory furniture.
What Is a Hospital Clinical Lab?
Hospital laboratories analyze human blood, saliva, urine, and other bodily fluids to support disease diagnosis, treatment monitoring, and prognosis assessment. Core departments include clinical chemistry, microbiology, biochemistry, immunology, HIV screening, and blood banking.
What testing equipment is used in hospital lab departments?
Testing equipment in hospital laboratory departments includes: biochemical analyzers, hematology analyzers, coagulation analyzers, urine sediment analyzers, centrifuges, water purifiers, printers, chemiluminescence analyzers, plate washers, microscopes, water baths, incubators,
refrigerators, biosafety cabinets, computers, and more.
What tests does a hospital lab perform?
The department’s primary function is to provide accurate and scientific testing data for clinical diagnosis, treatment monitoring, and prognosis assessment across various diseases, covering biochemistry, cytology, hematology, and immunology.
From pathogen identification to clinical drug susceptibility testing and bacterial resistance monitoring, the department provides recommendations for the rational use of antibiotics.

Core International Standards for Hospital Laboratory Design
Global laboratory design and operations adhere to recognized standards to ensure quality, biosafety, and compliance:
- ISO 15189: Medical laboratories—Quality and competence requirements
- ISO 15190: Medical laboratories—Safety requirements
- WHO Laboratory Biosafety Manual
- CLSI GP18 A2: Guidelines for Laboratory Design Accreditation
- Biosafety Levels (BSL): Standards for handling infectious materials
- SEFA 8: Performance requirements for laboratory furniture and workbenches
- BIFMA X5.9: Stability, load-bearing capacity, and safety requirements for laboratory storage systems
Core Functional Zones of a Clinical Laboratory
An efficient laboratory requires zoned operations to prevent cross-contamination and optimize workflows:
- Specimen reception and sorting area
- Emergency Testing Area
- Body Fluid Collection Area
- Instrument Room
- Clinical Biochemistry and Immunology Laboratory
- HIV Screening Laboratory
- Reagent Preparation Room
- PCR Laboratory
- Microbiology Laboratory
- Culture Room
- Cleanroom
A Comprehensive Planning and Design of Hospital Clinical Lab Facilities
The rationality of a hospital laboratory’s internal spatial design directly impacts workflows, internal communication, and operational efficiency. The overarching design principle is to reduce unnecessary physical barriers as much as possible and to design the laboratory environment as an aesthetically pleasing, spacious,
safe, comfortable, and efficient space.
The design must also fully account for plumbing and drainage systems, electrical systems, lighting, ventilation, and HVAC systems, as well as disinfection and infection control systems. The rational layout of the infrastructure for these systems directly impacts laboratory safety and may even affect the accuracy of test results.
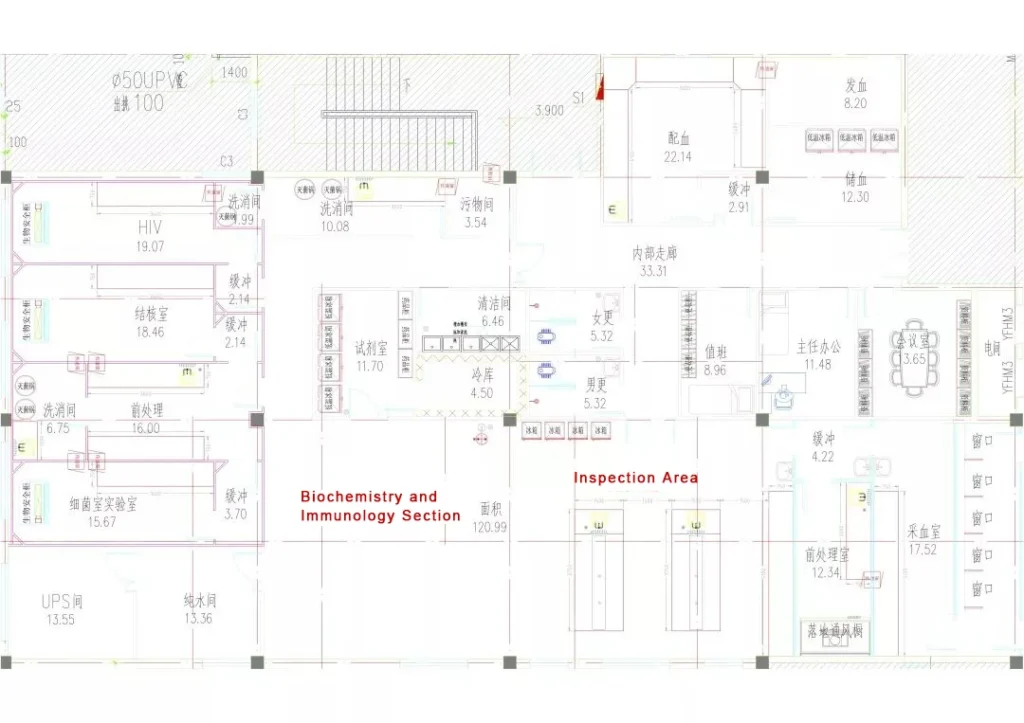
I. Floor Plan Layout and Zoning Requirements
The Clinical Laboratory Department should be located within the outpatient building as an independent area, with three major zones clearly physically separated:
- Contaminated Zone: Testing laboratories, phlebotomy room
- Semi-Contaminated Zone: Reagent storage, water treatment room, and other auxiliary functional areas
- Clean Zone: Offices, changing rooms
Personnel and material flows must be completely separated:
- Separate entrances and exits must be provided for staff, patients, specimens, and clean supplies
- Contaminated materials must have dedicated exits and waste elevators; sharing with passenger elevators is strictly prohibited
Specifications for Specialized Laboratory Rooms
- HIV Screening Laboratory: Clean / Semi-Contaminated / Contaminated zones, area ≥45 m²
- Microbiology Laboratory: Preparation room, buffer room, and work area; area ≥35 m²
- PCR Laboratory: Reagent preparation area → Sample preparation area → Amplification and analysis area; each area must have a buffer room; total area ≥60 m²
- Blood Collection Area: Separate partition; window length ≥1.2 m, width 45–60 cm
- Instrument Room: Separate pre-processing and instrument analysis areas; incompatible equipment must be housed in separate rooms
II. Interior Finishing and Cleanliness Standards
Finishing must meet infection control, easy-to-clean, and durable requirements:
- Floors: Seamless, slip-resistant, and corrosion-resistant; use rubber or PVC flooring; joints should be welded, ground, and polished to match the color of the flooring
- Walls and Ceilings: Easy to clean and disinfect, dust-free, crack-resistant, smooth, waterproof, and scrub-resistant; use double-sided sandwich-core color-coated steel panels; fire resistance rating ≥ Class B1 (flame-retardant)
- Doors: Self-closing, equipped with observation windows, door locks, and door closers; operational status indicator lights may be installed
- Windows: Openable exterior windows are not recommended; use sealed observation windows
- Joints: Wall-floor and wall-ceiling junctions should be rounded; color-coated steel panels should be sealed with sealant to ensure airtightness
- Ceiling Height: 2.6 m is recommended; maintenance access panels should not be installed in the main laboratory ceiling
III. Ventilation, Air Conditioning, and Airflow Control
Ventilation directly affects biosafety and specimen integrity:
- Clean areas shall have independent air handling units to prevent cross-contamination
- Design shall account for the thermal and humidity loads of biosafety cabinets, centrifuges, and incubators
- Three-stage filtration: Fresh air coarse filter → Medium-efficiency filter in the air handling unit → High-efficiency filter at the supply air outlet
- Fresh air inlets shall be ≥2.5 m above the floor, protected against rodents, insects, and rain, and equipped with easily removable and washable filters
- Exhaust and supply fans must be interlocked; exhaust fans should start before supply fans. Equipment exhaust and room exhaust must be separated into distinct circuits
- Directional airflow: Clean zone → Semi-contaminated zone → Contaminated zone, with a pressure differential of ≥5 Pa; install a visible pressure gauge
- Air conditioning components must be free of wooden materials, corrosion-resistant, and non-absorbent; air leakage rate < 2%
- Fan coil units + fresh air system; utilize the hospital’s centralized heating and cooling sources in winter and summer; provide air-cooled modular units for spring and autumn
IV. Electrical System
Reliable power supply to ensure core equipment operation and safety:
- Electrical load rating ≥ Class II; core equipment equipped with UPS, with a backup runtime of ≥ 30 minutes
- Adequate fixed outlets; critical equipment on separate circuits; equipped with ground fault circuit interrupters
- Lighting: Laboratories ≥ 300 lx; buffer rooms ≥ 200 lx; office areas ≥ 200 lx
- UV Sterilization Lamps: One 30W UV lamp per 10–15 m²
- Install evacuation signs, emergency lights, and exit signs in accordance with fire safety regulations
- Install network ports, access control systems, surveillance systems, and emergency call systems

V. Water Supply and Drainage Systems
- Pure Water System: Designed to meet the water requirements of biochemical analyzers; water usage points and volumes must be determined in advance
- Water Supply: Non-manual handwashing fixtures installed at outlets; piping made of stainless steel, copper, or non-toxic plastic
- Drainage: No floor drains in clean laboratories; laboratory and domestic wastewater must be separated and discharged to the hospital wastewater treatment plant
VI. Laboratory Furniture Requirements (Compliant with International Standards)
Laboratory furniture must address ergonomics, chemical resistance, and infection control, and comply with SEFA 8 and ISO 15189 standards:
Material Classification
- PP furniture (polypropylene): Resistant to strong acids and alkalis, it is manufactured using one-piece molding and seamless welding technology, which significantly enhances structural integrity.
- Stainless steel furniture: Robust, hygienic, durable, and corrosion-resistant in high-end manufacturing environments.
- Steel-wood furniture: Offers a balance of cost-effectiveness, strength, and hygiene.
- All-steel furniture: Highly durable and easy to disinfect.
Countertop Materials
- Recommended: Phenolic resin countertops, epoxy resin countertops, marble or ceramic countertops
- Characteristics: Chemical resistance, heat resistance, stain resistance, and ease of disinfection
- Avoid materials that are porous, easily damaged, or prone to microbial growth
Design & Installation Principles
- Dimensions Must Match Workflows, Equipment, and Space
- Adequate storage without overcrowding
- Sturdy and tip-resistant, with sealed edges and hardware
- Compliant with SEFA 8 requirements for chemical resistance and structural performance
- Meets infection control and ergonomic height standards
Conclusion
A scientifically planned hospital laboratory can improve diagnostic accuracy, staff safety, and operational efficiency while meeting strict international standards. From layout and airflow to furniture and finishes, every detail directly impacts laboratory operations and compliance.




